Kid AID Device
Standardised clinical data acquisition
Kid AID Device is the component of the project responsible for the safe, repeatable recording of short videos and observational parameters during routine paediatric examinations. The device operates in hospital ward and emergency department settings, ensuring a standardised process for collecting high-quality data.
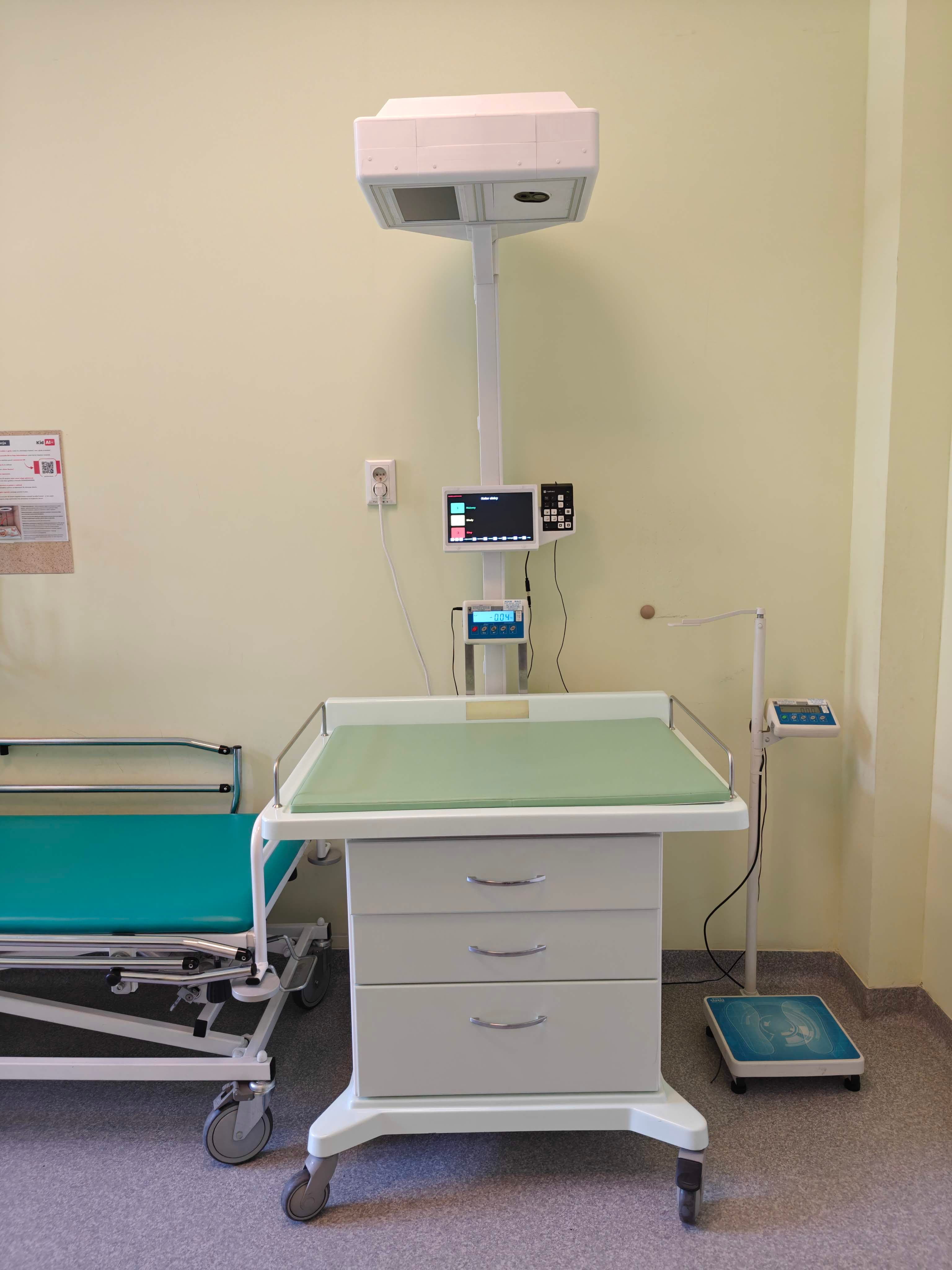
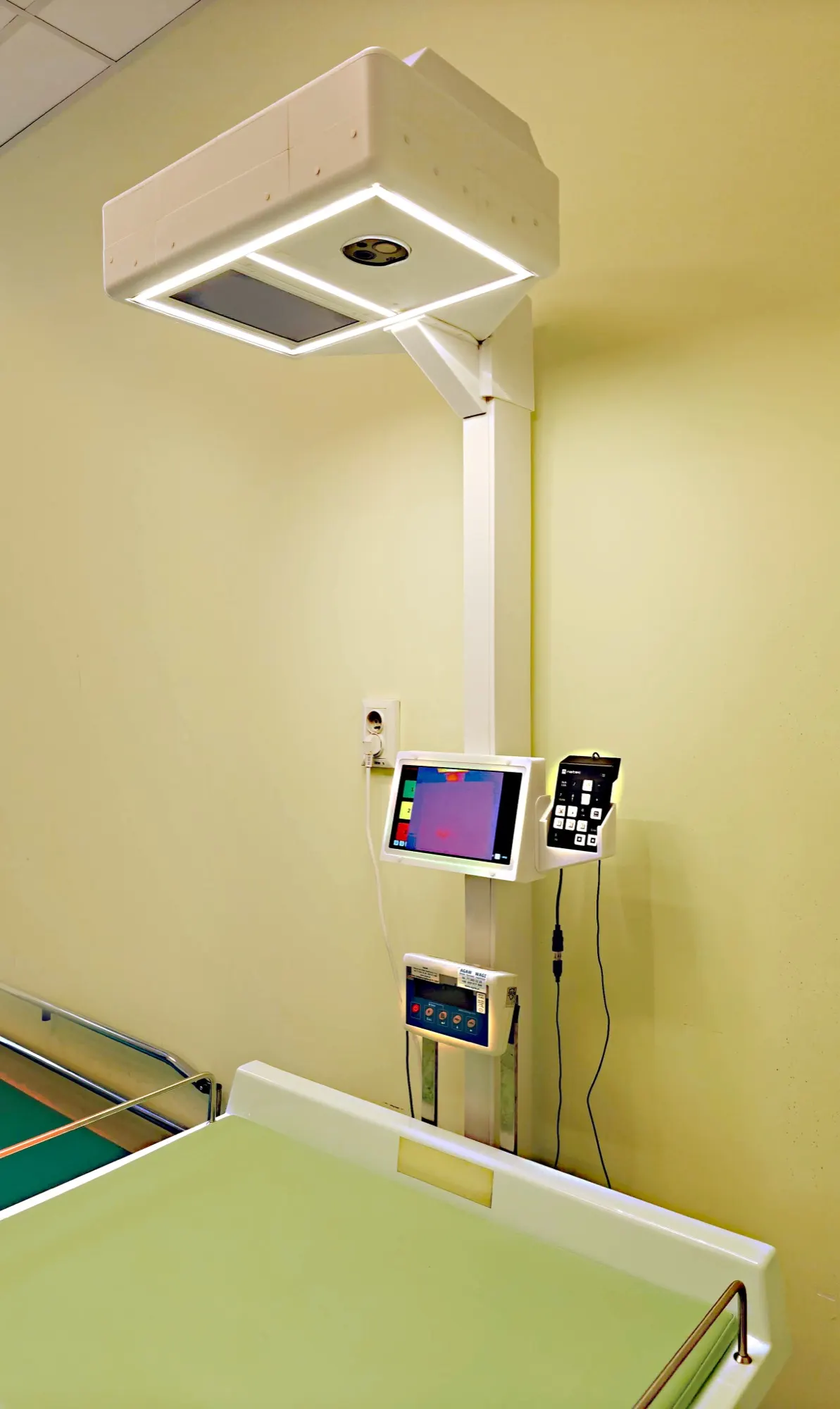
How does it work?
Recording lasts approximately 60 seconds
RGB image and thermal image are captured simultaneously
Recording is linked to complete clinical data
Recording is performed minimally – without affecting the examination
What does Kid AID Device analyse?
Data collected by Device forms the basis for developing AI algorithms and educational tools. The system integrates four layers of analysis.
Visual analysis (RGB camera)
Recording of the child's general appearance, skin colour, movement pattern, activity and response to the environment.
These are the parameters experienced paediatricians use to form the subjective assessment known as "ill appearance" (impression of a seriously ill child).
Thermal analysis (thermal camera)
Objective measurement of temperature distribution on the surface of the child's body.
Behavioural analysis
Observation of breathing pattern, spontaneous movements and child behaviour – parameters that in traditional examination depend on the doctor's subjective assessment.
Photoplethysmography analysis
Photoplethysmography (PPG) in Kid AID enables non-contact assessment of peripheral perfusion and circulatory parameters of the child based on analysis of skin colour changes in the video recording. The method allows extraction of heart rate and microcirculation quality information without touching the patient.
Clinical data integration
Each recording is associated with a complete set of medical information:
- Medical history and symptoms
- Laboratory and imaging results (if performed)
- Medical interventions taken
- Disease follow-up and discharge summary
Technical specification
- RGB camera
- High-quality video recording
- Thermal camera
- Measurement of body surface temperature distribution
- Recording duration
- approx. 60 seconds
- Working environment
- Emergency department / SOR / paediatric ward
- Data security
- Pseudonymisation, encryption, access control
Goal – data, not diagnosis
Kid AID Device does not make a diagnosis or take clinical decisions. Its role is to supply high-quality data on which the next two components of the Kid AID system are built:
Clinical centres
Data are collected in three clinical centres in Lower Silesia:

Department of Paediatrics and Infectious Diseases, Wrocław Medical University
Leading centre, scientific coordination

St. Hedwig of Silesia Hospital in Trzebnica
Partner centre

Dr A. Sokołowski Specialist Hospital in Wałbrzych
Partner centre
Planned expansion: Centres in Warsaw (Paediatric Emergency Department of the Paediatric Hospital, Medical University of Warsaw) and other Polish cities, as well as international centres.
Safety and ethics
The project is carried out in accordance with the highest ethical and legal standards for the protection of medical data.
- Voluntary participation – requires consent from legal guardians
- Approval of the UMW Bioethics Committee
- GDPR compliance – pseudonymisation and encryption
- Data minimisation – we collect only essential information